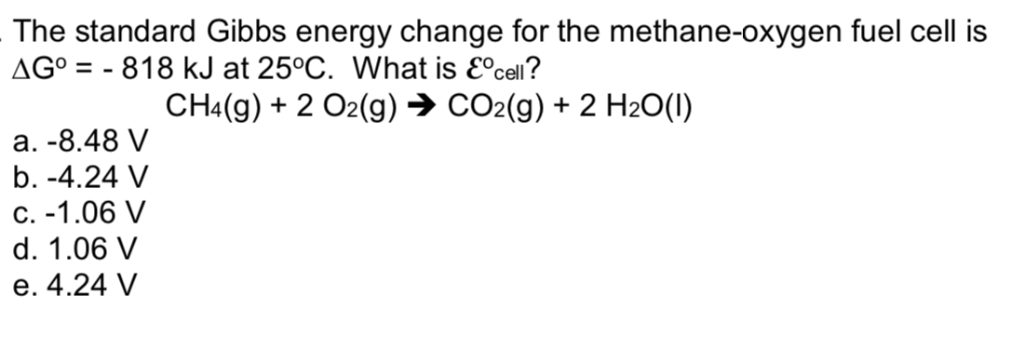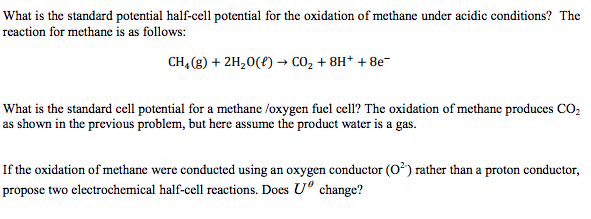Fcevs also present relatively low barriers to entry in terms of societal changes as they operate and perform similarly to conventional vehicles refuelling at stations in minutes and driving for 500 to 600 kilometres on a single tank.
Ch4 fuel cell.
The material catalyzes an oxidation process that releases carbon dioxide electrons and protons.
Fuel cell hydrogen oxygen fuel cell hydrocarbon oxygen fuel cell 12 csbe electrochemistry duration.
Similar to today s gasoline vehicles fuel cell electric cars can have a driving range of more than 300 miles on one tank of hydrogen fuel.
And cationic ions fe 3 and cu 2 resulting from the corrosion of fuel cell stack system components.
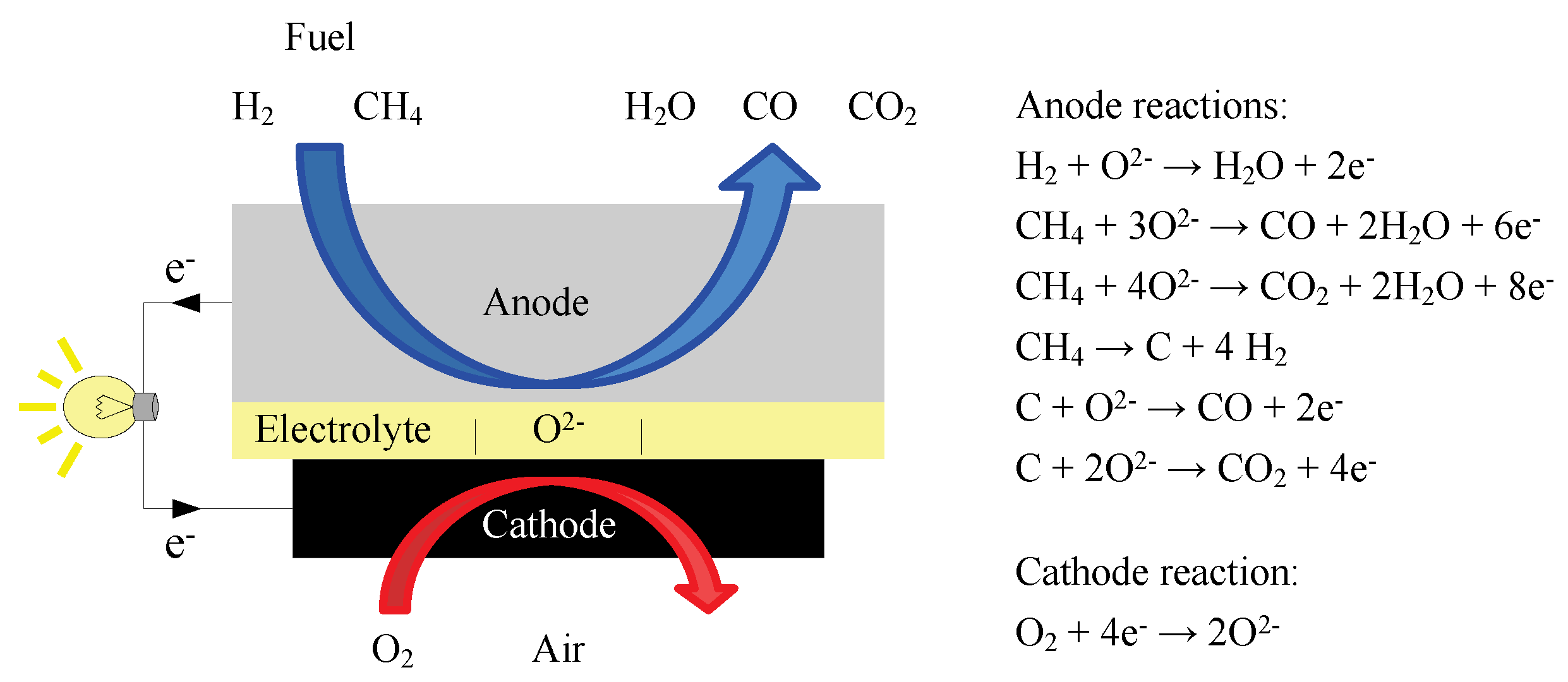
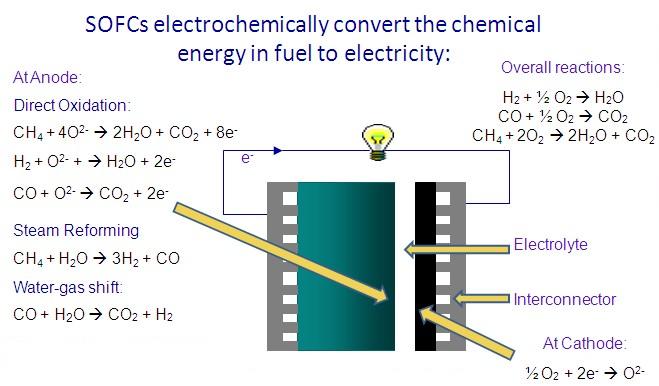
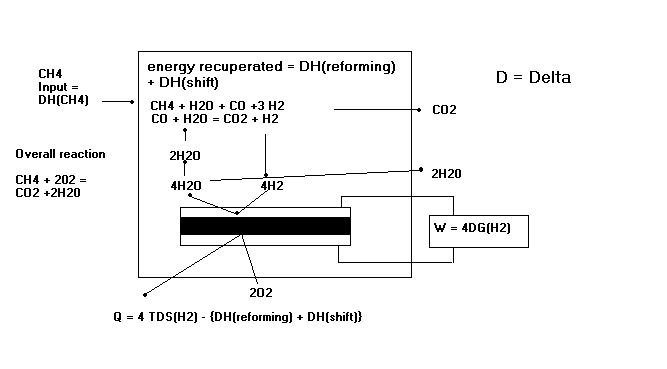
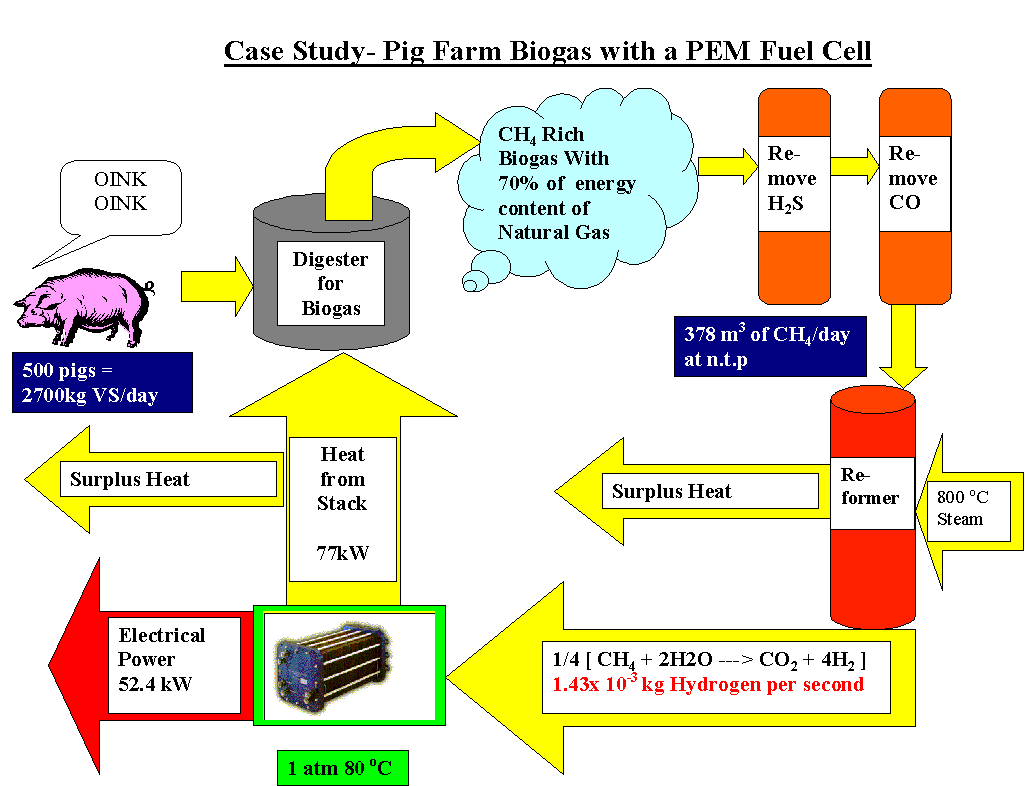
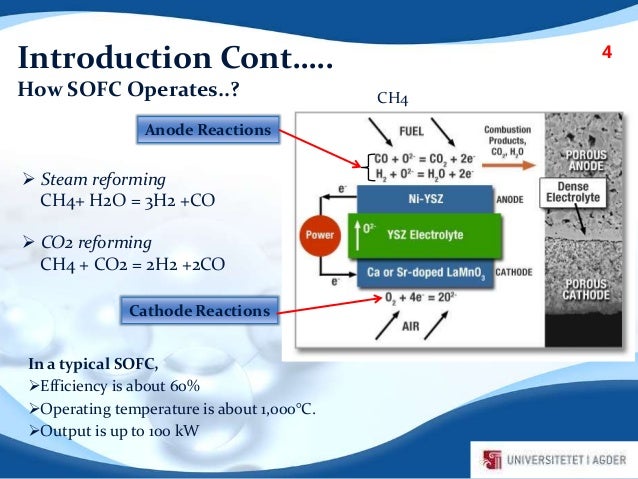
Methane is first reformed to co and h2 within the fuel cell and then the h2 and co are electrochemically oxidized to h2o and co2 on the electrode the lower temperature fuel cell would allow ordinary stainless steel rather than exotic materials to be used for the interconnectors that link the cells into a stack.
Air pollutants no x so x co and co 2.
The team covalently attached a low temperature methane activating platinum catalyst to conductive carbon and used the material as a fuel cell anode.
It was found that even trace amounts of impurities present in either fuel.
This is much cleaner than octane and more efficient.
The contaminants included were fuel impurities co co 2 h 2 s and nh 3.
The delta g is 818 kj.
A robust fuel cell operated on nearly dry methane at 500 c enabled by synergistic thermal catalysis and electrocatalysis.
I don t get how to get the n value to plug into delta g nfe.
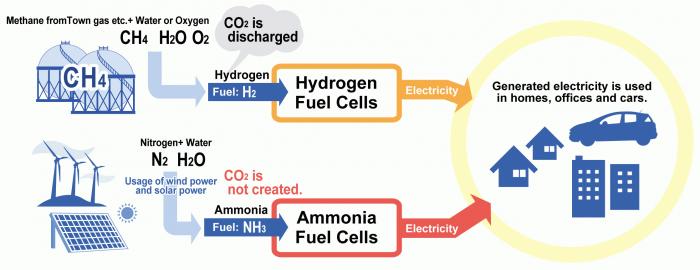
Take methane and make a fuel cell that adds oxygen to the ch4 and generate electricity 4 water molecules and 1 carbon dioxide molecule.
A fuel cell by contrast can mix hydrogen with air to produce electricity at up to 60 per cent efficiency.
6 methane natural gas fuel cell.
Learners planet 75 443 views.
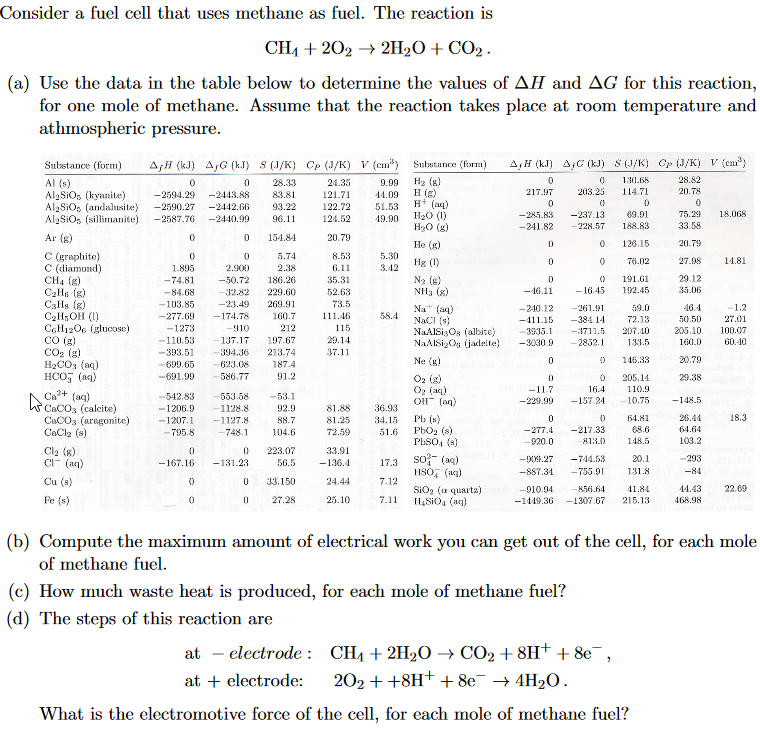
Consider a fuel cell that uses methane as fuel.
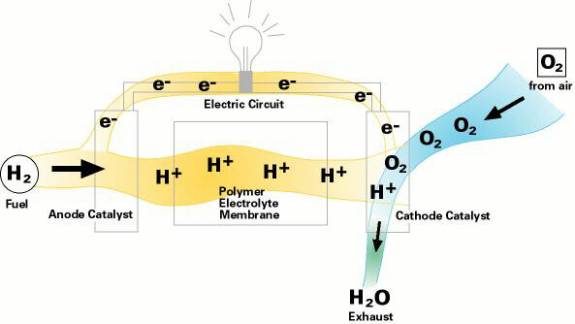
The protons then react with oxygen at the cathode to produce water.
Fuel cell cars are very similar to traditional gasoline powered cars.
This paper reviewed over 150 articles on the subject of the effect of contamination on pem fuel cell.
Consider a fuel cell that uses the reaction ch4 g 2o2 g co2 g 2h2o l given the standard free energies of the formation in appendix b what is the value of e.